‘Breakthrough’ drugs under scrutiny
An influential Alzheimer’s drugs report has concluded that so-called “breakthrough” treatments are unlikely to provide meaningful benefits to patients.
Researchers said the impact of the drugs was “well below” what is needed to significantly improve the lives of people living with dementia.
How the drugs work
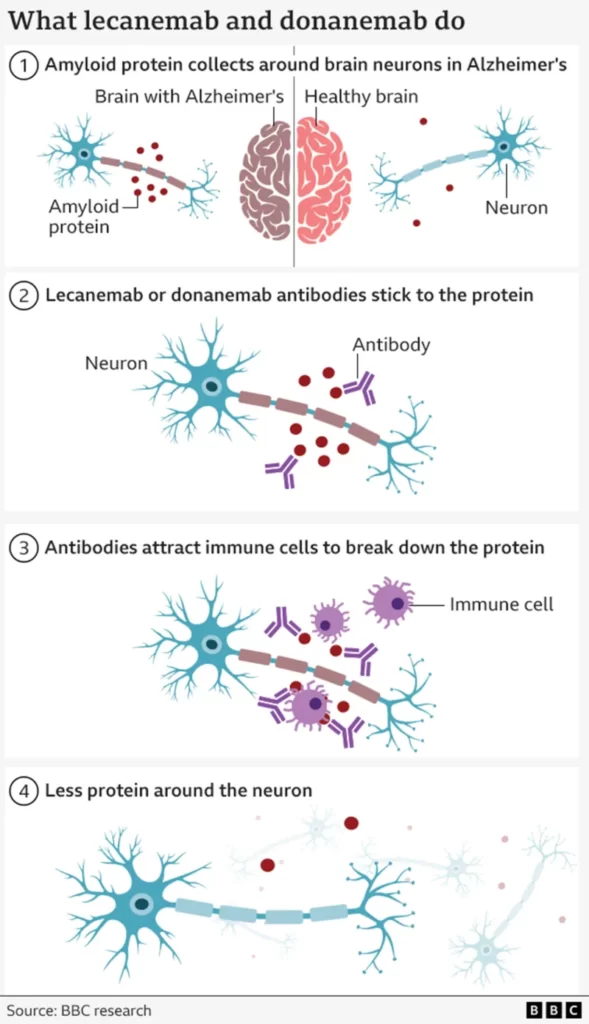
The treatments target beta amyloid, a sticky substance that builds up between brain cells in Alzheimer’s disease.
Engineered antibodies are designed to identify and remove this amyloid from the brain. While this approach failed for years, newer drugs such as donanemab and lecanemab have shown they can slow cognitive decline.
This marked the first time any drug has slowed brain deterioration linked to Alzheimer’s.
Findings from large-scale analysis
The Cochrane Collaboration analysed 17 studies involving more than 20,000 participants.
The Alzheimer’s drugs report found that while the treatments do slow disease progression, the effect is too small to make a meaningful difference for patients.
The drugs also carry risks, including brain swelling and bleeding, and require frequent administration every two to four weeks, at a high financial cost.
Concerns over expectations and cost
Experts involved in the analysis stressed the importance of managing patient expectations.
They warned against offering false hope, noting that the treatments may place a burden on both patients and their families without delivering substantial benefits.
Currently, access to these drugs is limited, with patients in the UK required to pay privately, making them unaffordable for most.
Scientific backlash and debate
The report has sparked strong criticism from other scientists, who argue that the analysis is flawed.
Some experts say it unfairly groups older, less effective drugs with newer treatments that have shown measurable, though modest, benefits.
Critics argue that this approach obscures progress made in recent years and risks undermining decades of research.
Ongoing review and future directions
The National Institute for Health and Care Excellence has previously declined to approve these drugs for NHS use but is reviewing the evidence again.
Researchers say alternative approaches, such as targeting brain inflammation, should now be explored as part of future Alzheimer’s treatment strategies.
Source: adapted from information by the BBC
Also read: Huntington’s disease treatment gives patients new hope
For more videos and updates, check out our YouTube channel


